
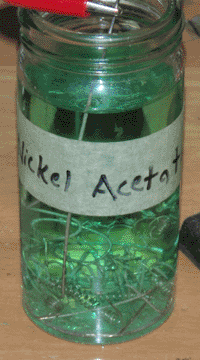

Nickel is used often in the construction vacuum tubes. It spot welds readily to most metals, it can withstand high temperatures without sagging or evaporating, and it can easily be soldered to. It follows then, that in experimenting with vacuum tubes, nickel scrap (from failed tubes and wire clippings) would accumulate. To reuse this scrap, I developed a simple method to plate copper with a durable coat of nickel.
I started with a small jar of vinegar (5% acetic acid) to which I added the majority of my scrap nickel wire, which sank to the bottom. I then added two long pieces of nickel as electrodes, one of which touched the scrap pile. I connected my DC power supply across the two electrodes, with the positive side being the scrap pile. I let this run at 12 volts for about three hours, until the solution had turned a light green and crystals of nickel had begun to grow on the negative electrode. At this point the plating solution (nickel acetate) was ready.
To plate the piece of copper shown above, I first filed and sanded off the oxide layer, then stirred it in vinegar to remove any remaining oxide and to slightly etch the surface. I then suspended it in the plating solution and connected it to the negative side of my DC power supply, which was set to 3.2 volts; the positive end was connected to the scrap nickel in the solution. The copper turned a silver color immediately, but I let it continue plating for about 3 minutes after that to build up a thicker coat. Finally, I turned off the power supply and removed the copper from the solution.
Through trial and error I have found that the time limit for this method of nickel plating is around 30 minutes. Beyond this point the coat becomes irregular and thick crystalline scales begin to form, which are mechanically weak and tend to flake off. Up until this point however, the nickel coat is very durable, and even thin coats will not wipe off unless scraped by a sharp metal edge.